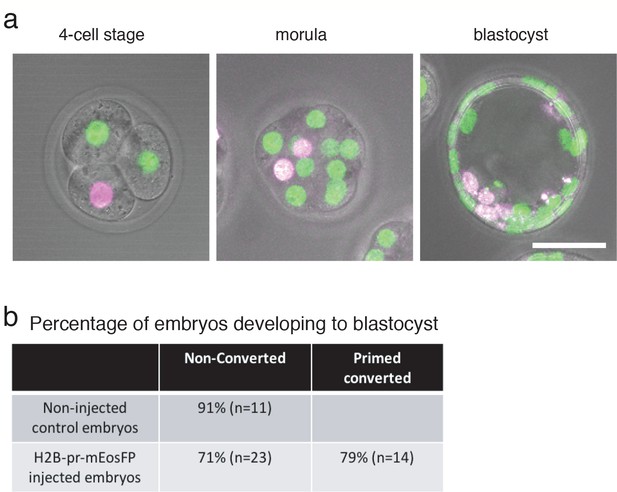
Selective plane illumination microscopy (SPIM) has the potential to play a major role in achieving comprehensive, non-invasive imaging of mammalian pre-implantation development. This should help scientists look into how early life processes give rise to specialized cells, and even explore the fate of cells in other tissues.Īccurate lineage tracing and precise tracking of single cells in pre-implantation embryos are essential for a mechanistic understanding of the first cell fate decisions during mammalian development ( Welling et al., 2016 Pantazis and Bollenbach, 2012). The new approach means the experiments require fewer imaging events, but also fewer embryos because even the ones that move a lot can be studied. designed a piece of software, baptized ‘primed Track’, that can use this red cell (and its daughters) to reorient the embryo during image analysis and reliably identify and match any mother cell to its daughters. Then, a technique known as primed conversion was used so that only one cell in a four-cell embryo would glow red. made embryonic cells produce a special fluorescent marker, which is normally green but can be converted to red. To address this problem, Welling, Mohr et al. Taking images more frequently is not possible because each imaging event exposes the embryo to light, which can damage its fragile cells. However, many embryos move and turn around between images, and so software packages often lose track of which cell was which. 
This helps reconstruct the history of each cell, and which structures they give rise to. Images are taken regularly and then processed by specialized software that automatically tracks the fluorescent cells and their daughters over time. These embryos are grown inside a special microscope for several days. To do so, researchers genetically manipulate embryos so that each cell produces fluorescent molecules that ‘glow’ under light. Scientists want to track these early events to understand how they give rise to an individual formed of huge numbers of cells organized in specialized tissues. Within three to four days, certain cells begin to specialize and take on specific roles. Using green and red labels as fiducial markers, we computationally correct for rotational and translational drift, reduce overall data size, and accomplish high-fidelity lineage tracing even for increased imaging time intervals – addressing major concerns in the field of volumetric embryo imaging.Ī mouse embryo starts with one cell, which divides to create identical daughters that quickly start to multiply. Confined primed conversion of H2B-pr-mEosFP-labeled nuclei combined with light-sheet imaging greatly facilitates segmentation, classification, and tracking of individual nuclei from the 4-cell stage up to the blastocyst. Here, we exploit the advantageous properties of primed convertible fluorescent proteins (pr-pcFPs) to simultaneously visualize the global green and the photoconverted red population in order to minimize tracking uncertainties over prolonged time windows. Lineage tracing using global fluorescence labeling techniques is complicated by increasing cell density and rapid embryo rotation, which hampers automatic alignment and accurate cell tracking of obtained four-dimensional imaging data sets. Accurate lineage reconstruction of mammalian pre-implantation development is essential for inferring the earliest cell fate decisions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
